 Values for the standard molar entropy, in J/mol Using this reference, the standard molar entropy, S°, is the entropy of 1 mole of a substance under standard state conditions. Second, all entropy values can be measured against a fixed reference point-the entropy at absolute zero. There are two major consequences of the third law of thermodynamics.įirst, at temperatures greater than absolute zero, the entropy of all substances must be positive. Solving Boltzmann’s equation, the entropy is equal to zero. Thus, these components have a singular microstate, and W is equal to 1. The third law of thermodynamics states that at zero Kelvin, also known as absolute zero, the entropy of a pure, perfectly crystalline substance is zero.Īt zero Kelvin, the components of a crystal have no kinetic energy and no molecular motion, meaning that they can only occupy one fixed position. With greater molecular motion, a substance has more ways to distribute the kinetic energy among its components that is, it has a greater number of possible microstates. 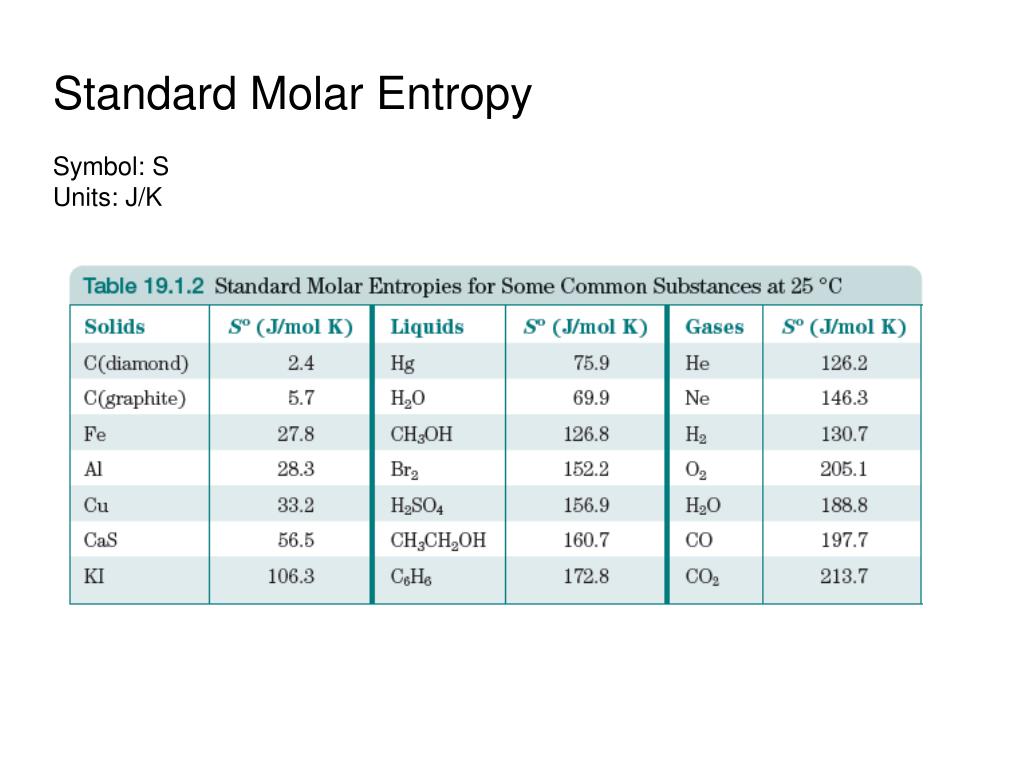
If gloves are worn, PVA, butyl rubber and viton are suitable materials.The components of a substance have kinetic energy, which appears as different types of molecular motion, including translational, rotational, and vibrational motion. Instead, retain in a safe place in the laboratory (well away from any source of ignition)įor disposal with other flammable, non-chlorinated solvents. It is probable that trying to dispose of benzene in this way will also break localĮnvironmental rules. It is dangerous to try to dispose of benzene by washing it down a sink, since it is toxic, will cause environmental damageĪnd presents a fire risk. If swallowed: Call for immediate medical help. Reddens or appears damaged, call for medical aid. Skin contact: Wash off with soap and water. Wear safety glasses and use protective gloves.Įye contact: Immediately flush the eye with plenty of water. If benzene must be used in an experiment, it should be handled at all stages in a fume cupboard. The pure material, and any solutions containing it, constitute a fire risk.īenzene should NOT be used at all unless no safer alternatives are available. *** Benzene is a carcinogen (cancer-causing agent). Uses formula: log e P m m H g = obtained from CHERIC Note: yellow area is the region where the formula disagrees with tabulated data above.ĭistillation data Vapor-liquid Equilibrium Note: (s) notation indicates equilibrium temperature of vapor over solid, otherwise value is equilibrium temperature of vapor over liquid. Table data obtained from CRC Handbook of Chemistry and Physics 44th ed. /GettyImages-98831556-56a134d03df78cf7726861a5.jpg)
Structure and properties Structure and properties It is highly recommended to seek the Material Safety Datasheet ( MSDS) for this chemical from a reliable source such as SIRI, and follow its directions. The handling of this chemical may incur notable safety precautions. This page provides supplementary chemical data on benzene. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
